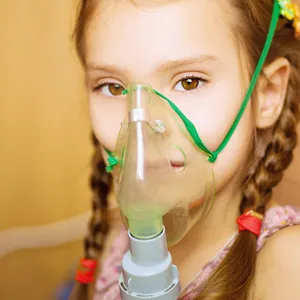
Jess Nickless, from Devon, has asked the government to make Kalydeco available as a treatment for patients with the rare G551D mutation of cystic fibrosis, a serious genetic disorder that causes damage to the lungs and the digestive system.
Approved by the European Medicines Agency, Kalydeco is available in the US and Italy. Its listed cost is £182,000 per patient per year.
But while the European Medicines Agency advises that Kalydeco only be prescribed to patients over the age of six, Ms. Nickless, whose daughter Ellie has G551D cystic fibrosis, wants it to be available to patients of all ages. Citing the fact that her daughter has already sustained lung damage due to the disease, Nickless believes that waiting until her daughter is six for treatment with the drug will put her at greater risk.
For her campaign, Nickless has launched an e-petition that currently has more than 11,000 signatures.
Her efforts are being supported by another mother of a child with the G551D mutation, Denise Murphy, who calls it a “travesty” that the UK is behind the US, Germany and Italy in terms of the availability of Kalydeco.
According to the DoH, decisions on funding for Kalydeco are up to local NHS chapters.
Source: BBC


